
Anodes in electroplating: what they are and their function in the process
The critical component for industrial electrochemical stability
What is an anode in electroplating?
In technical terms, the anode is the positive electrode in an electrolytic cell. In industrial electroplating, the anode is not just a passive component; it is the raw material in solid form that “dissolves” in a controlled manner to replenish the metal concentration of the bath as the metal is deposited onto the parts.

How anodes work in the electroplating process
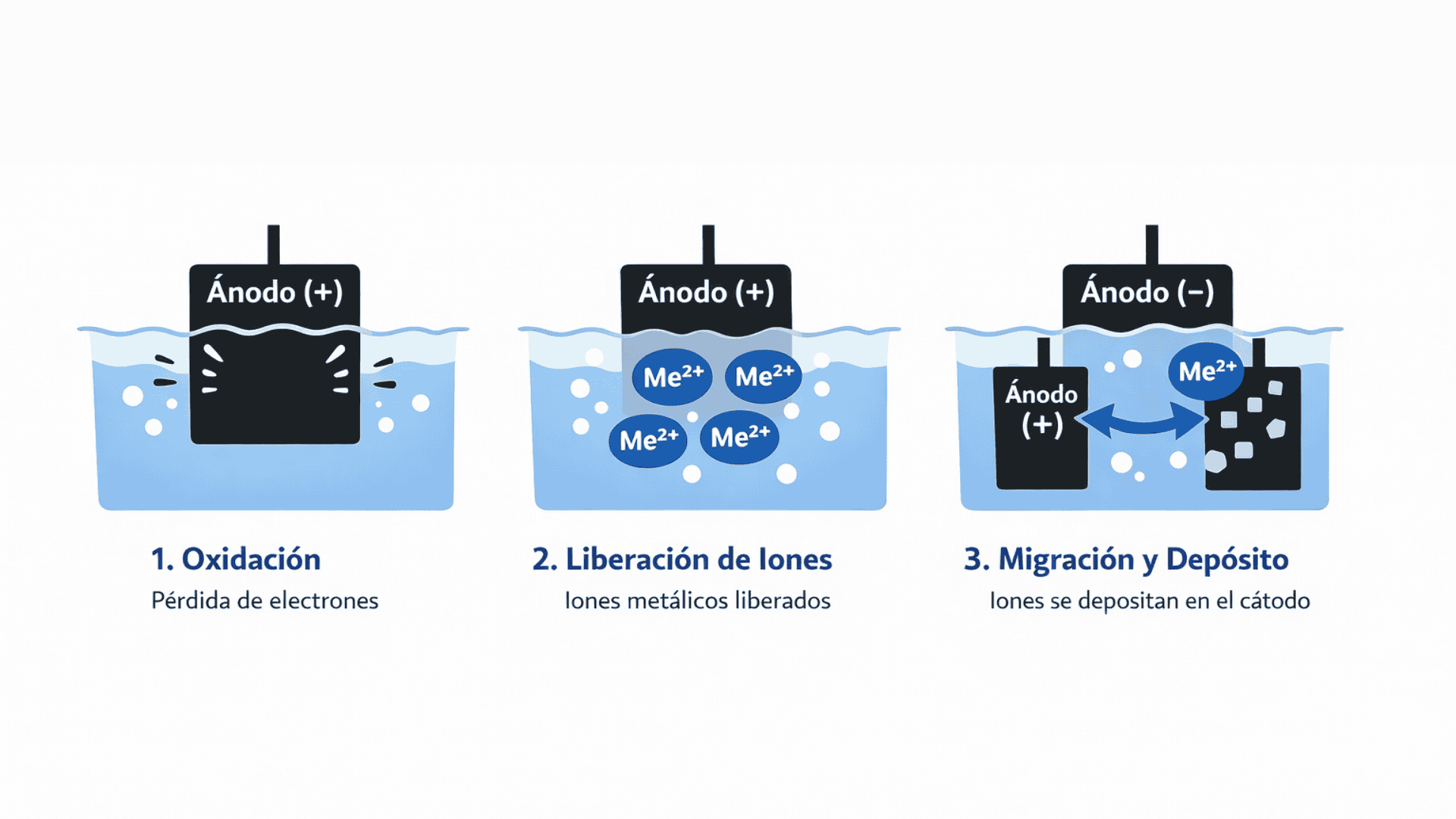
The operation can be summarized in three critical stages:
-
Oxidation: Under electric current, metal atoms on the surface of the anode lose electrons.
-
Ion release: These atoms become positively charged ions that enter the solution (electrolyte).
-
Migration and deposition: The ions travel to the cathode (the part), where they regain electrons and crystallize as a solid, uniform coating.
Process efficiency depends on the geometry and composition of the anode. At Conalsol, we manufacture tailored solutions:
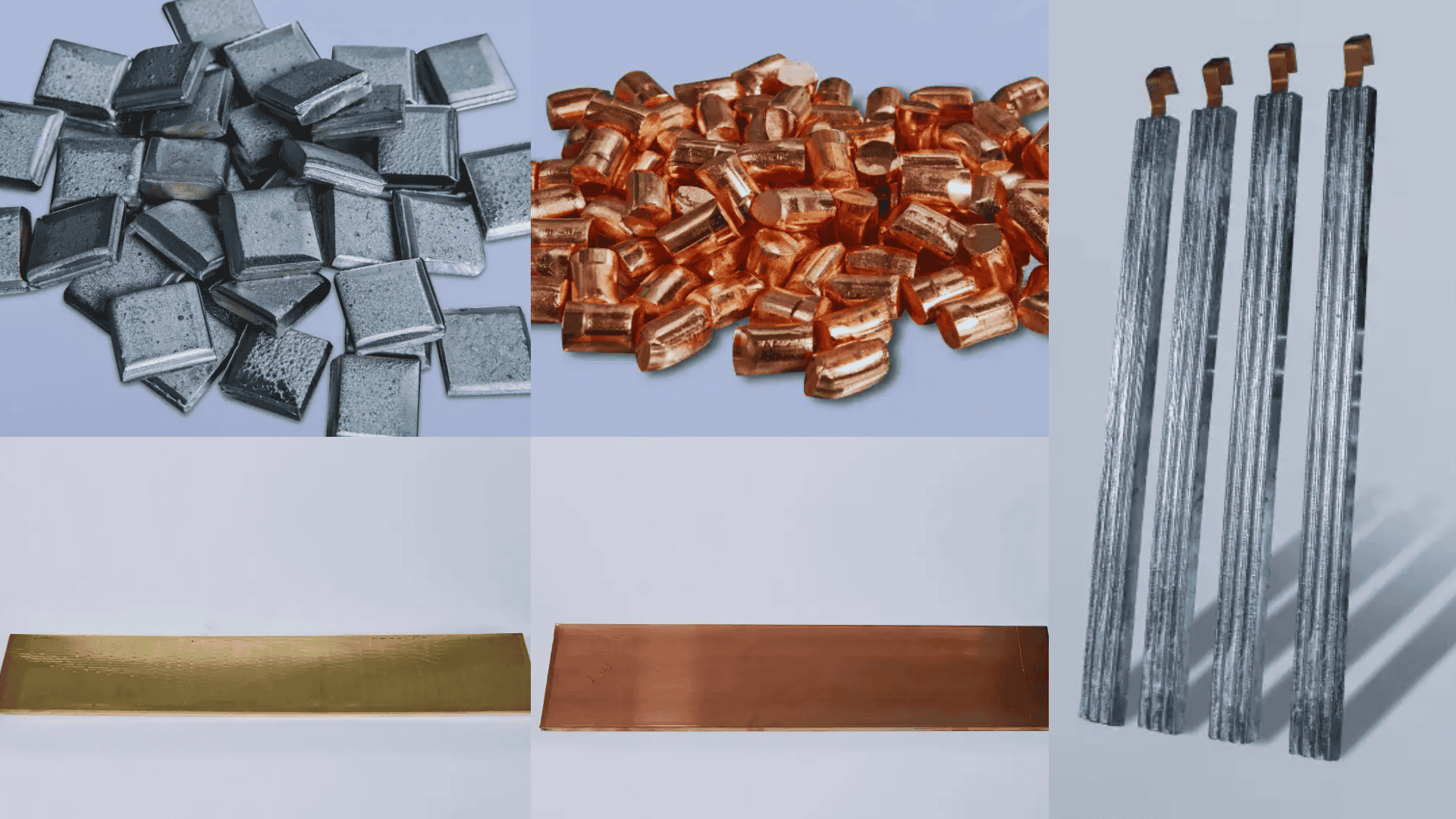
Zinc anodes:
Essential for electrogalvanizing. We offer high-purity electrolytic zinc to prevent sludge formation at the bottom of the tank.
Copper anodes:
Available in plates and phosphorized mini-cylinders for baskets, ideal for processes requiring fast and consistent dissolution.
Tin anodes:
Manufactured in pure tin or tin-lead alloys, ensuring superior protection and solderability.
Nickel anodes:
Available in 1×1 and 4×4 squares, designed for maximum contact surface.
Lead and brass anodes:
Ideal for chrome plating processes (as conductors) or specific decorative finishes.
Importance of anodes in coating quality
A common mistake in plant management is to consider the anode as a “commodity” based only on price. However, its impact is directly reflected in:
-
Metal purity: An anode with 0.1% contamination can unbalance a 5,000-liter bath in weeks.
-
Uniform dissolution: Prevents the formation of “spikes” or anode passivation, which would cause voltage drops.
-
Bath stability: A good anode maintains constant metal concentration, reducing the need to add costly chemical salts.
Difference between anodes and coating metals
It is essential not to confuse their roles: while coating metals define the final properties perceived by the customer, electroplating anodes are the technical tool that makes that coating possible.
In electroplating processes, the metals used in coatings determine the final properties of the part. If you want to learn which are the most commonly used and their industrial applications, you can read our article on metals used in electroplating.
Conclusion

Choosing the right anode is ultimately a decision about the operational stability of your plant. A high-quality anode from Conalsol does not just supply metal—it delivers predictability, reduces waste, and ensures that the Quality Engineer can certify each batch with confidence.
Do you need a specific alloy or anode geometry? At Conalsol, we manufacture custom anodes with or without copper hooks. Contact us today for personalized technical advice.
Recibe asesoría personalizada
¿Listo para comenzar a proteger tu inversión en cada milla náutica?
