
Most commonly used metals in electroplating and their industrial applications
Properties, selection criteria, and performance in modern industry
In today’s industrial ecosystem, the competitiveness of a component lies not only in its geometric design or base alloy, but in the integrity of its surface. Industrial metal coatings are the critical barrier that separates functionality from premature failure.
Electroplating, through technical electrodeposition, enables the transfer of properties from noble or resistant metals to more economical or lightweight substrates, optimizing the cost-benefit ratio of production.
From corrosion resistance in saline environments to conductivity in microelectronics, the choice of sacrificial or coating metal determines the asset’s service life. In this article, we will analyze the most commonly used metals in electroplating and how to select the right one according to industry requirements.
What is electroplating?
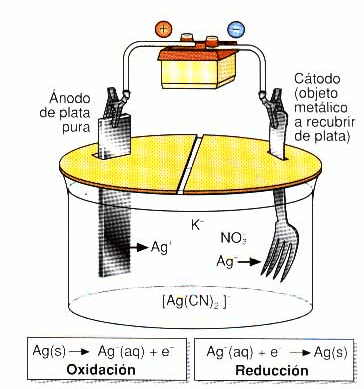
Electroplating is a precision electrochemical process in which direct current is used to transfer metal ions from an anode (the metal source) to a cathode (the part to be coated and protected), both immersed in a conductive solution called an electrolyte.
This metal electrodeposition phenomenon allows the creation of nanometric or micrometric layers with atomic-level adhesion—something impossible to achieve with conventional paints or mechanical coatings.
Why is choosing the right metal important?
For a Procurement Manager or Quality Engineer, selecting the metal is not an aesthetic detail—it is a financial and operational decision. Each metal provides a specific technical “personality” to the component:
-
Corrosion resistance: Galvanic or barrier protection
-
Electrical conductivity: Crucial to prevent hot spots in connectors
-
Wear resistance: Increased surface hardness (Vickers scale)
-
Solderability: Facilitates subsequent assembly processes
Most commonly used metals in electroplating
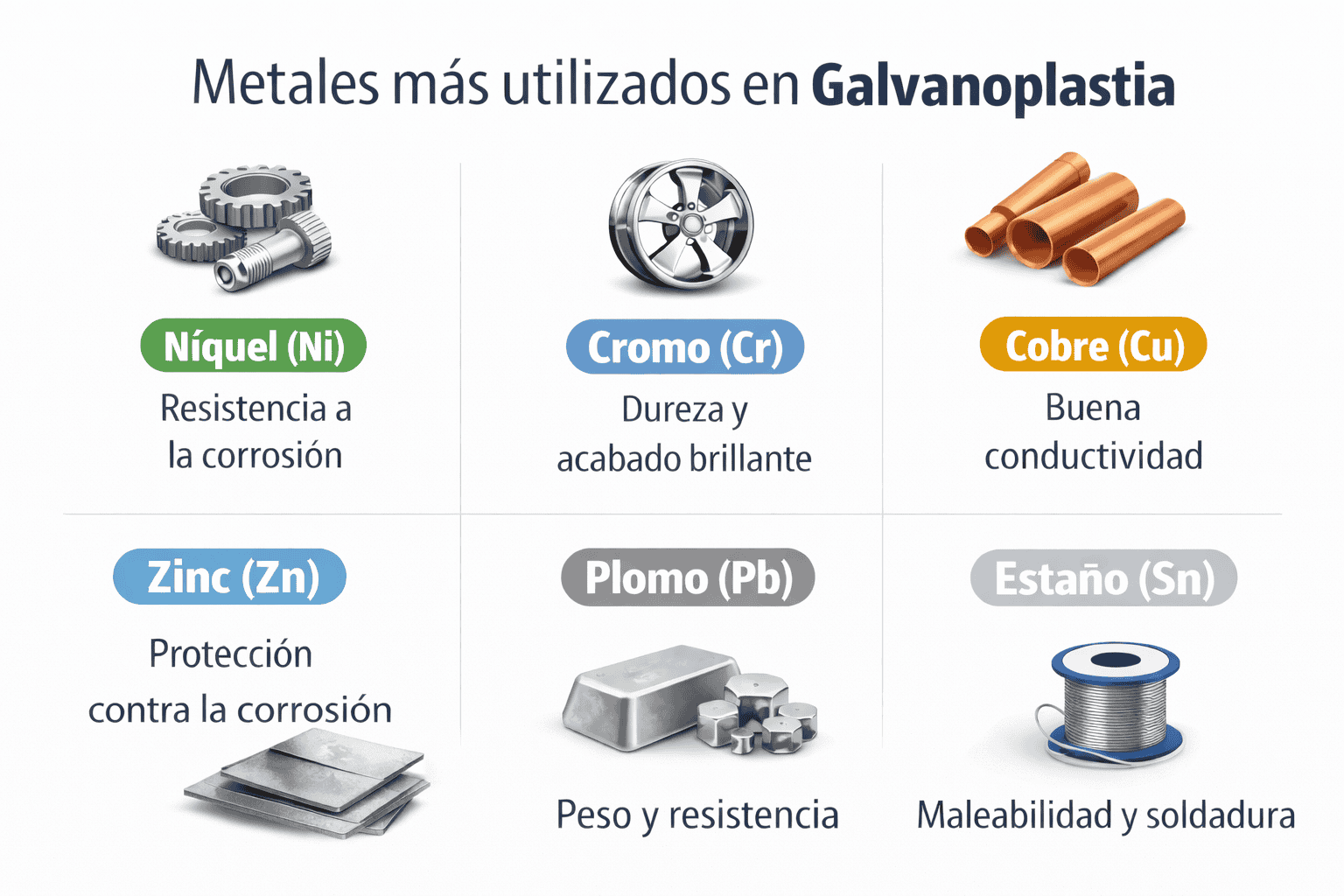
Zinc in electroplating: The protection standard
Zinc is the sacrificial metal par excellence. Its main function is the galvanic protection of steel, where zinc corrodes before the base metal.
-
Properties: Excellent cost-to-protection ratio
-
Applications: Fasteners, construction structures, and automotive chassis components
Nickel in electroplating: Hardness and aesthetics
Nickel offers a unique combination of chemical resistance and a bright finish. It is essential as an intermediate layer in multi-metal processes.
-
Properties: High hardness, wear resistance, and surface leveling capability
-
Uses: High-end faucets, industrial machinery components, and hand tools
Copper in electroplating: The backbone of adhesion
Copper is irreplaceable due to its superior thermal and electrical conductivity, as well as its role as a “bridge layer” that ensures adhesion of other metals.
-
Properties: Extreme ductility and thermal conductivity
-
Uses: Printed circuit boards (PCBs), power connectors, and electrodes
Lead and its alloys: Resistance in chemical environments
Given Conalsol’s specialization in non-ferrous metals, lead stands out where others fail, especially in the chemical and energy storage industries.
-
Properties: Exceptional resistance to sulfuric acid and high density for radiation shielding
-
Uses: Chemical tank linings, specialized sacrificial anodes, and battery components
Tin in electroplating: Safety and solderability
Tin is valued for its low toxicity and excellent solderability, making it essential in regulated industries.
-
Properties: Resistance to nitriding and food-grade compatibility
-
Uses: Food canning industry (tinplate), electronic terminals, and electrical contacts
Comparative table of metals used in electroplating
| Metal | Main Property | Key Industry | Critical Function |
|---|---|---|---|
| Zinc | Galvanic protection | Construction / Automotive | Cost-effective corrosion protection |
| Nickel | Hardness and shine | Machinery / Decorative | Wear resistance |
| Copper | Conductivity | Electronics | Adhesion base |
| Tin | Solderability | Electronics / Packaging | Stable electrical contact |
| Lead | Chemical inertness | Chemical processes | Acid resistance |
Property: Galvanic protection
Industry: Construction / Automotive
Function: Cost-effective corrosion protection
Property: Hardness and shine
Industry: Machinery / Decorative
Function: Wear resistance
Property: Conductivity
Industry: Electronics
Function: Adhesion base
Property: Solderability
Industry: Electronics / Packaging
Function: Stable electrical contact
Property: Chemical inertness
Industry: Chemical processes
Function: Acid resistance
| Metal | Main Property | Key Industry | Critical Function |
|---|---|---|---|
| Zinc | Galvanic protection | Construction / Automotive | Cost-effective anti-corrosion |
| Nickel | Hardness and shine | Machinery / Decorative | Wear resistance |
| Copper | Conductivity | Electronics | Adhesion base |
| Tin | Solderability | Electronics / Packaging | Stable electrical contact |
| Lead | Chemical inertness | Chemical processes | Acid resistance |
The origin of quality: The anode
It is essential to understand that, in industrial electroplating, the final metal coating on the part comes directly from the anode. The anode is the material that releases metal ions into the electrolyte under the influence of electric current.
Anodes play a fundamental role in the electroplating process, as they are the pure source of the deposited metal. If you want to learn more about their technical function and specialized types, check out our article on Anodes in electroplating: what they are and their function.
Conclusion
To achieve efficient electroplating processes, it is essential to use high-quality metals and materials that guarantee consistent results. Marginal savings in initial metal purity translate into exponential costs due to quality rejection.
At Conalsol, we supply non-ferrous metals with certifications that ensure the stability of your plating bath from the very first moment.
FAQ
1. What metals are used in electroplating?
The most common are Zinc, Nickel, Copper, Tin, Lead, Gold, and Silver.
2. What is the most widely used metal?
Zinc, due to its extensive use in preventing steel corrosion.
3. What is the difference between galvanizing and electroplating?
Galvanizing typically refers to hot-dip coating in molten zinc, while electroplating is a more precise and versatile electrochemical process.
Recibe asesoría personalizada
¿Listo para comenzar a proteger tu inversión en cada milla náutica?
